Why does HCl dissociate in water?
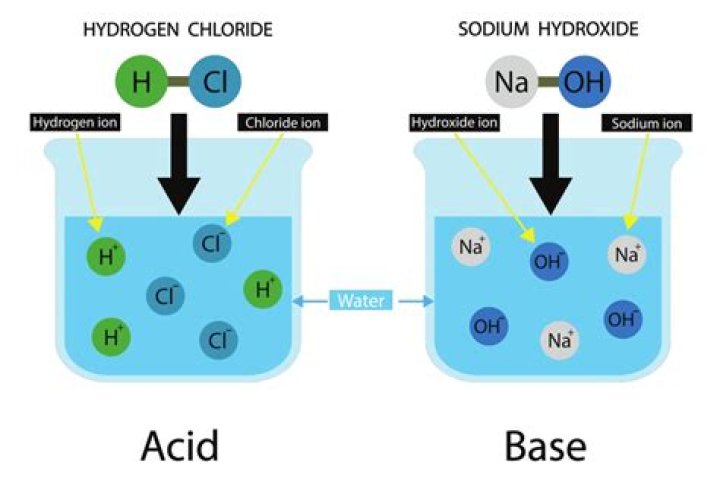
HCl will dissociate in water because HCl is also a polar molecule (Cl is slightly negative and H is slightly positive). The Cl will be attracted to the slightly positive H in water and the H will be attracted to the slightly negative O in water.
Will HCl fully dissociate in water?
Dissociation of acids When HCl molecules dissolve they dissociate into H+ ions and Cl- ions. HCl is a strong acid because it dissociates almost completely.
What is the equation for the dissolution of HCl in water?
When it dissolves in water, it produces positive hydrogen ions and negative chloride ions (Cl-). This can be represented by the chemical equation: HCl H2O→ H+ + Cl.
Does HCl dissociate in aqueous solution?
We can calculate that in 1 mol/L HCl there are more than 1200 H₃O⁺ ions for every molecule of undissociated HCl. For all practical purposes, HCl is completely dissociated in solution. Strong acids have a large dissociation constant, so they dissociate completely in water.
What is the dissociation constant of HCl?
Because hydrochloric acid, HCl(aq), is a strong monoprotic acid, the value for its acid dissociation constant, Ka, would be extremely large….pH = -log10[0.10] = 1.
| %dissociation = | [H+] [HCl initial] | × 100 |
|---|---|---|
| %dissociation = | 0.1 0.1 | × 100 |
| %dissociation = | 100% |
Does HCl dissociate or ionize?
Hydrogen chloride (HCl) ionizes completely into hydrogen ions and chloride ions in water. A weak acid is an acid that ionizes only slightly in an aqueous solution.
Why do some acids dissociate completely?
They are strong electrolytes. The hydrogen atom that is lost during dissociation is *not* strongly bound to the rest of the acid molecule. Therefore, the solvent (usually water) pulls at the H+ atom more strongly than the rest of the acid molecule.
What is HCl formula?
HCl
Hydrochloric acid/Formula
What happens to HCl in water?
HCl is a strong electrolyte and when it dissolves in water it separates almost completely into positively – charged hydrogen ions and negatively – charged chloride ions. This aqueous solution is usually called hydrochloric acid.
Does HCl have equilibrium constant?
(Note: Equilibrium constants are truly constant only at a given temperature, pressure, and solvent condition. For a irreversible reaction, such as the reaction of a 0.1 M HCl(aq) in water, [HCl]eq = 0, so you can’t easily measure a Keq. …
What is the general dissociation reaction of acid?
Definitions. According to Arrhenius’s original molecular definition, an acid is a substance that dissociates in aqueous solution, releasing the hydrogen ion H+ (a proton): HA ⇌ A− + H+. The equilibrium constant for this dissociation reaction is known as a dissociation constant.
How does HCl ionize?
A strong acid is an acid which is completely ionized in an aqueous solution. Hydrogen chloride (HCl) ionizes completely into hydrogen ions and chloride ions in water. A weak acid is an acid that ionizes only slightly in an aqueous solution. Because HCl is a strong acid, its conjugate base (Cl − ) is extremely weak.