What will happen the boiling point of water if 5 g of salt is added into it?
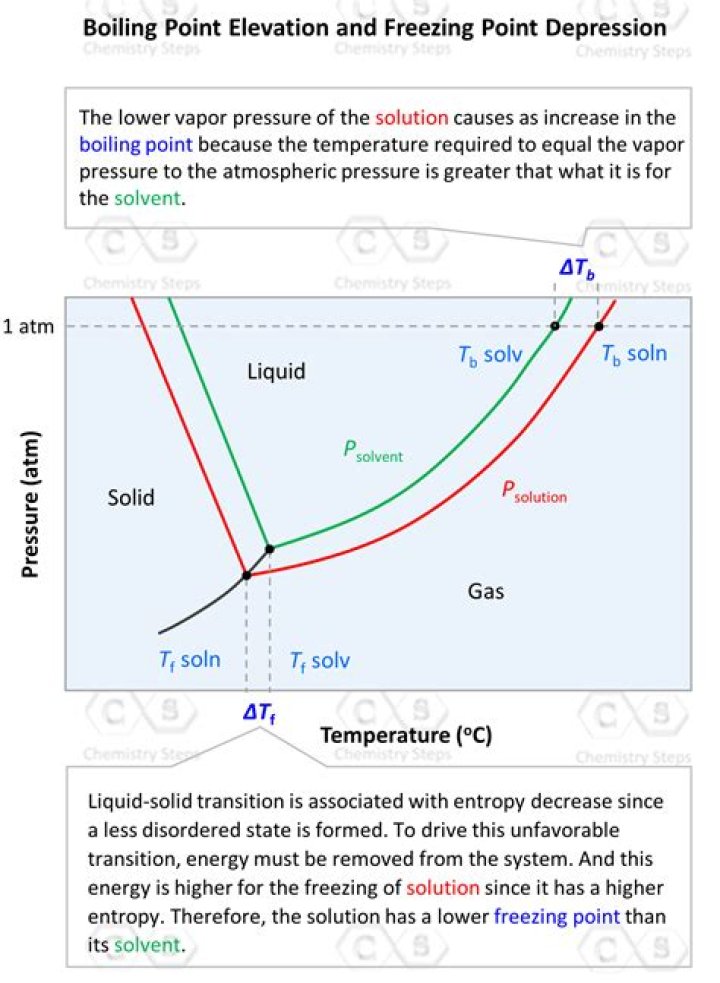
When table salt is added to water, the resulting solution has a higher boiling point than the water did by itself. The ions form an attraction with the solvent particles that prevents the water molecules from going into the gas phase. Therefore, the saltwater solution will not boil at 100oC.
How much salt will raise the boiling point of water?
So yes, salt increases the boiling temperature, but not by very much. If you add 20 grams of salt to five litres of water, instead of boiling at 100° C, it’ll boil at 100.04° C. So a big spoon of salt in a pot of water will increase the boiling point by four hundredths of a degree!
What happens to the boiling temperature of water as salt is added?
If you add salt to water, you raise the water’s boiling point, or the temperature at which it will boil. The temperature needed to boil will increase about 0.5 C for every 58 grams of dissolved salt per kilogram of water. This is an example of boiling point elevation, and it is not exclusive to water.
What is the relationship between tablespoons of salt and boiling temperature?
6) Use thermometer to record the highest temperature of the boiling saltwater. VI: ANALYSIS: My data table and graph clearly shows that adding salt raises the boiling temperature of water. For every tablespoon of salt added, the boiling temperature increased by almost 3 degrees.
Do you add salt before or after boiling water?
Add the salt when the water comes to a boil. Wait until the water returns to a full, rolling boil before putting in the pasta.”
Does salt in water make it boil faster?
One particularly stubborn myth is that adding salt will make the water take longer to come to a boil. Chemically speaking, it’s true that salt raises the boiling point; however, the amount of salt used in cooking applications is so small that it won’t make a difference with timing.
What happens to the salt solution after boiling?
When salt is added, it makes it harder for the water molecules to escape from the pot and enter the gas phase, which happens when water boils, Giddings said.
Can the salt be separated out using this distillation system?
For example, water can be separated from salt solution by simple distillation. This method works because water has a much lower boiling point than salt.
Why add salt after water boils?
When salt is added, it makes it harder for the water molecules to escape from the pot and enter the gas phase, which happens when water boils, Giddings said. This gives salt water a higher boiling point, she said.
Do you add salt when boiling potatoes?
Boil the Potatoes Add ½ to 1 teaspoon salt to the water. Cook the potatoes in gently boiling water until tender, about 15 minutes for small Red Potatoes, New Potatoes or cubed large potatoes, and 20 to 25 minutes for quartered potatoes.
How does table salt affect the boiling temperature of water?
Beaker/ Concentration of Nasal (M) Boiling Point Temperature Pure tap water 100. (00 1 tablespoon salt solution 102 (CO) tablespoon salt solution 103. 5 (CO) 3 tablespoon salt solution 105 (CO) Graph the data from Table 2 Label each axis (X and Y will NOT do). Devise a scale for each axis and label that scale (1, 2, 3…
How to boil tap water with table salt?
Kitchen stove or electric hot plate 2. One 2-cup measuring cup 3. Measuring spoon set, with one tablespoon maximum 4. Table salt 5. Tap water 6. 2 quart cooking Pot (1/2) i don’t have sorry 7. One digital thermometer Procedure (list steps in the order in which you performed them): 1. Boil one quart of tap water on stove. .
What happens when you add salt to water?
Many inorganic dissolved solids increase the conductivity Salt elevates the boiling temperature of water using a process known as boiling point elevation.
What is the boiling point of 2 h tablespoons of water?
Use your graph to determine the boiling point of a solution made with 2 h tablespoons. This use of a graph has generated a hypothesis that a 2 h tablespoon solution boils at 104 degrees C. Prepare a 2. 5 M solution and verify your hypothesis.
What should the boiling temperature of water be when you add salt?
Measure the temperature of the water once it gets to a boil. You should get something around 212 degrees F although it may vary slightly. Then, measure out one tablespoon of salt. Pour it into the boiling water and stir it well. Use the thermometer again to take the temperature of the water and record the finding.
How much salt would one need to add 1 kg water to change?
In reality, you would need to add 230 grams of table salt to a liter of water just to raise the boiling point by 2° C. That is 58 grams per half degree Celsius for each liter or kilogram of water. That is much more salt than anyone would care to have in their food.
How does dissolved salt affect the temperature of water?
The temperature will increase about a one-half degree Celsius for every 58 grams of dissolved salt per kilogram of water. This is an example of boiling point elevation. The property isn’t exclusive to water.
Updated June 28, 2019. If you add salt to water, you increase its boiling point. The temperature will increase about a one-half degree Celsius for every 58 grams of dissolved salt per kilogram of water.