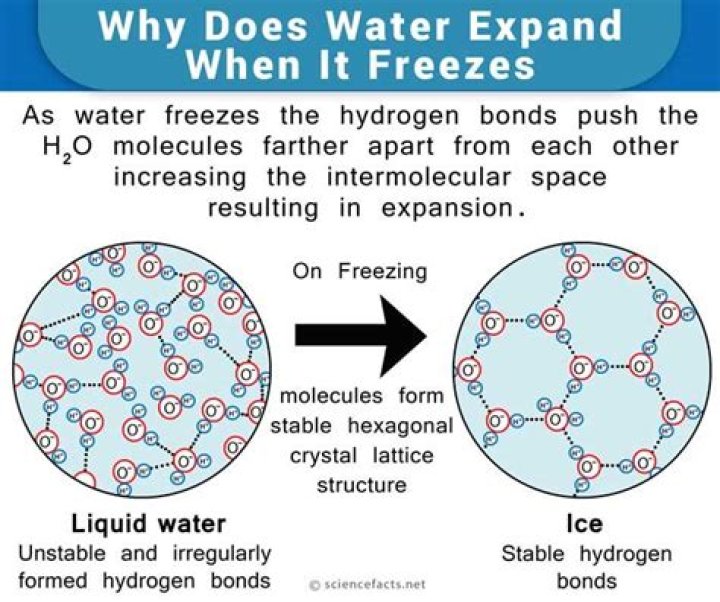
What happens to water molecules when they freeze?
Point out that when water freezes, the water molecules have slowed down enough that their attractions arrange them into fixed positions. Water molecules freeze in a hexagonal pattern and the molecules are further apart than they were in liquid water. Note: The molecules in ice would be vibrating.
What can break the hydrogen bonds between water molecules?
Hydrogen bonds are not strong bonds, but they make the water molecules stick together. The bonds cause the water molecules to associate strongly with one another. But these bonds can be broken by simply adding another substance to the water. How does a piece of ice float on top of water?
Do hydrogen bonds break in ice?
The hydrogen bonds between water molecules in ice produce the open structure shown in the figure below. When ice melts, some of these bonds are broken, and this structure collapses to form a liquid that is about 10% denser. This unusual property of water has several important consequences.
Why does water freeze in the freezer?
Molecules are always moving. Freezing happens when the molecules of a liquid get so cold that they slow down enough to hook onto each other, forming a solid crystal. For pure water, this happens at 32 degrees Fahrenheit, and unlike most other solids, ice expands and is actually less dense than water.
How does molecular motion change during the freezing process?
When you remove heat from water (or any substance), the molecules slow down. The freezing process occurs when the molecules stop sliding and tumbling all over each other (liquid phase) and settle into fixed positions in a large network called a crystal lattice, which is the solid phase.
How many hydrogen bonds are in ice?
four hydrogen bonds
Thereby, two atoms may act as H donors, two as H acceptors. In ice, as opposed to water clusters or liquid water, all four hydrogen bonds are developed, resulting in a tetrahedral coordination around each water molecule.
Why are the hydrogen bonds in liquid water less stable than ice?
There is less movement of molecules in colder conditions. The hydrogen bonds in liquid water are less stable than in ice. In ice, the water molecules are farther apart than in liquid water. when water freezes, ice floats.
What bonds water molecules together in ice?
The Case of H2O In ice, the crystalline lattice is dominated by a regular array of hydrogen bonds which space the water molecules farther apart than they are in liquid water. In other words, the presence of hydrogen bonds enables ice to float, because this spacing causes ice to be less dense than liquid water.
Why do hydrogen bonds expand in ice?
During freezing, water molecules lose energy and do not vibrate or move around as vigorously. This allows more stable hydrogen-bonds to form between water molecules, as there is less energy to break the bonds. Thus water expands as it freezes, and ice floats atop water.
Why does water under ice not freeze?
Because ice is a crystal, which means it has a regular pattern with spaces in between molecules. The spaces in the crystal are larger than the spaces between molecules in the liquid. More space = less dense, so ice is less dense than water. So although the air is freezing cold, the water isn’t freezing yet.
Why did my water freeze when I took it out of the freezer?
Why does this happen? It is because the water in the bottle is supercooled. A supercooled liquid is one in which the temperature is below its normal freezing point, but the liquid has not solidified. Another trigger can be the shock wave generated by hitting a bottle just out of the freezer against a table.
Why does water turn to ice when it freezes?
As the liquid cools down, the amount of potential energy is reduced and the molecules start to move slower. When the water temperature reaches around 0°C, the molecules stick together and form a solid – ice.
What happens to hydrogen bonds when water freezes?
And at temperatures low enough to turn off the disruptive effects of thermal motions, water freezes into ice in which the hydrogen bonds form a rigid and stable network. Notice that the hydrogen bond (shown by the dashed green line) is somewhat longer than the covalent O—H bond.
What type of bond is formed between two water molecules?
In the case of water, hydrogen bonds form between neighboring hydrogen and oxygen atoms of adjacent water molecules. The attraction between individual water molecules creates a bond known as a hydrogen bond.
Why does the density of water increase when it freezes?
Localized clusters of hydrogen bonds still remain, however; these are continually breaking and reforming as the thermal motions jiggle and shove the individual molecules. As the temperature of the water is raised above freezing, the extent and lifetimes of these clusters diminish, so the density of the water increases.
What type of Bond makes water sticky?
Cohesion: Hydrogen Bonds Make Water Sticky. Water has an amazing ability to adhere (stick) to itself and to other substances. The property of cohesion describes the ability of water molecules to be attracted to other water molecules, which allows water to be a “sticky” liquid.