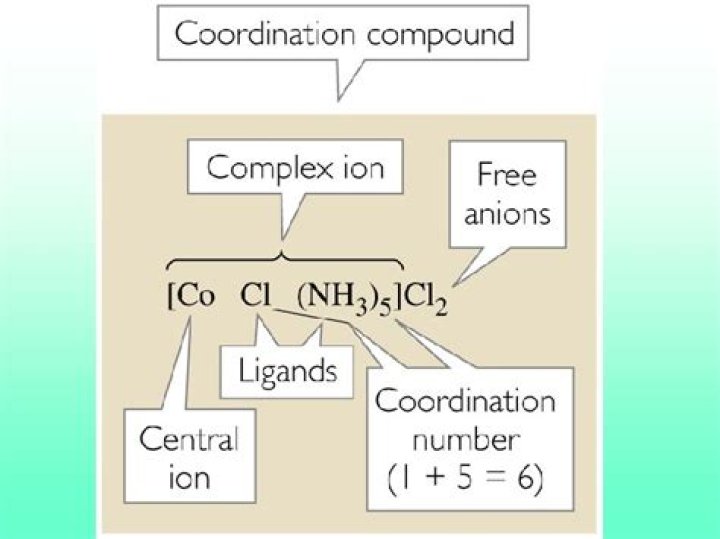
How do you write the formula of a coordination compound?
The formula of such complex compounds are written as per of the following rules: (i) Central atoms is written first. (ii) It is followed by the symbols of the ligands which are written in the order-negative ligands, neutral ligands followed by positive ligands.
How are complex designated?
If the complex ion is a cation, the metal is named same as the element. For example, Co in a complex cation is called cobalt and Pt is called platinum. (See examples 1-4.) If the complex ion is an anion, the name of the metal ends with the suffix -ate.
How do you name complexes with bridging ligands?
Bridging ligands are those bound to more than one central atom. They are differentiated in names by the addition of the prefix ‘μ’ (Greek mu), with the prefix and the name of the bridging ligand being separated from each other, and from the rest of the name, by hyphens.
What is the name of NCS?
N-Chlorosuccinimide
| Names | |
|---|---|
| CAS Number | 128-09-6 |
| 3D model (JSmol) | Interactive image |
| Abbreviations | NCS |
| Beilstein Reference | 113915 |
How do you write complex formulas?
Writing the (Line) Formula of a Complex:
- Identify the central metal ion.
- Identify the oxidation state on the central metal ion (shown in Roman numerals parantheses)
- Identify the ligands.
- Identify the number of ligands.
- Calculate the total charge on the ligands.
- Calculate the charge on the complex ion.
What is the Iupac name of k3 Al c2o4 3?
potassium aluminium (III) oxalate.
How do you write Iupac name?
In summary, the name of the compound is written out with the substituents in alphabetical order followed by the base name (derived from the number of carbons in the parent chain). Commas are used between numbers and dashes are used between letters and numbers. There are no spaces in the name.
How are coordination compounds named?
A. To name a coordination compound, no matter whether the complex ion is the cation or the anion, always name the cation before the anion. (This is just like naming an ionic compound.)
How do you find the coordination number of coordination compounds?
Here are the steps for identifying the coordination number of a coordination compound.
- Identify the central atom in the chemical formula.
- Locate the atom, molecule, or ion nearest the central metal atom.
- Add the number of atoms of the nearest atom/molecule/ions.
- Find the total number of nearest atoms.
How do you name chelating ligands?
Ligands with two or more points of attachment to metal atoms are called chelating ligands, and the compounds they make are called chelates, a name derived from the Greek word khele, or claw of a crab. Non-chelating ligands such as ammonia (NH3) are monodentate, with one point of attachment (one tooth).
How do you name complex inorganic compounds?
When naming a complex ion, the ligands are named before the metal ion. Write the names of the ligands in the following order: neutral, negative, positive. If there are multiple ligands of the same charge type, they are named in alphabetical order.
What are the IUPAC rules of Nomenclature of coordination compounds?
IUPAC Rules of Nomenclature of Coordination Compounds Anion Symbol Ligand name Acetate C H 3 C O O – acetato Crabonate C O 3 2 – carbonate Nitrate N O 3 – Nitrato Nitrite O N O – Nitro or nitrito – O
What are the rules for naming complex compounds?
In order to name complex compounds certain rules have been framed by IUPAC. These are as follows : (1) The positive part of a coordination compound is named first and is followed by the name of negative part. (2) The ligands are named first followed by the central metal.
How do you write the name of a coordination compound?
In a coordination compound, first, the name of the cation is written, followed by the name of the complex ion. If there is an anion, its name is written in the last. In naming a complex ion, first, the names of the ligands are written, followed by the name of the central metal atom or metal ion.
How do you name coordination compounds with neutral ligands?
The following neutral ligands are assigned specific names in coordination compounds: NH3 (ammine), H2O (aqua or aquo), CO (carbonyl), NO (nitrosyl). After the ligands are named, the name of the central metal atom is written. If the complex has an anionic charge associated with it, the suffix ‘-ate’ is applied.